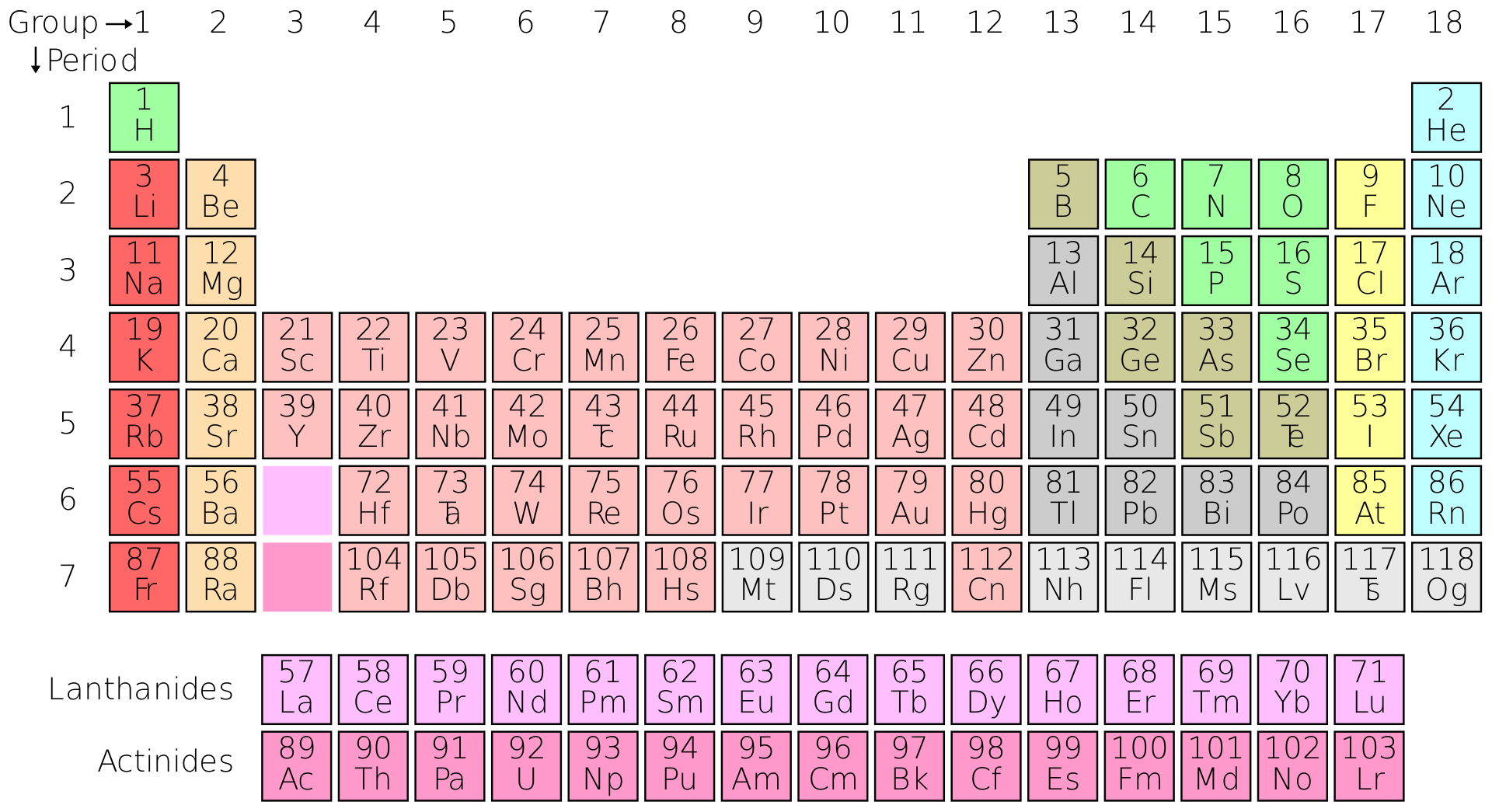
 note 1 Zinc is the 24th most abundant element in Earth's crust and has five stable isotopes. In some respects, zinc is chemically similar to magnesium: both elements exhibit only one normal oxidation state (+2), and the Zn 2+ and Mg 2+ ions are of similar size. Notice how Na after in the second I.E, Mg in the third I.E., Al in the fourth I.E. It is the first element in group 12 (IIB) of the periodic table. Zinc electronic configuration is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10. These are the ionization energies for the period three elements. Zinc is an element number 30 from transition metals family. Visualize trends, 3D orbitals, isotopes, and mix compounds. Therefore, it requires less energy to remove one of their valence electrons Table 1: Ionization Energies of certain elements (1st IE, 2nd IE, etc) Element Interactive periodic table showing names, electrons, and oxidation states. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in Zn (OH)2: Molar Mass (g/mol) Zn (Zinc) 1 × 65.409 65.409. Additionally, elements in the left corner have a low ionization energy because losing an electron allows them to have the noble gas configuration. 
The highest amount of energy required occurs with the elements in the upper right hand corner. IUPAC Periodic Table of the Elements and Isotopes (IPTEI) 9.3 Isotopes Used as a Source of Radioactive Isotope(s) The 68 Zn (p, 2p) 67 Cu (with a half-life of 62 h) reaction in which targets with zinc enriched in 68 Zn are irradiated and the neutron induced reaction 67 Zn (n, p) 67 Cu are both processes for producing 67 Cu for radiotherapy 254. The highest ionization energies are the noble gases because they all have high effective charge due to their octet formation and require a high amount of energy to destroy that stable configuration. The original periodic table of the elements published by Dimitri Mendeleev in 1869 arranged the elements that were known at the time in order of increasing atomic weight, since this was prior to. Ionization energies increase relative to high effective charge. One molecule of water (H 2 O) would weigh 18.02 amu (2×1.00797 amu for H + 15.9994 amu for O), and a mole of water molecules would weigh 18.02 grams. (The above picture of the periodic system is interactive - no need to download, just click on an element.\nonumber \] Now available: history of the periodic tableĬhoose elements by name, by atomic number, by symbol, by massĬlick here for the history of the periodic table.Ĭlick here to download a PDF version from that periodic table An interactive, printable extended version of the Periodic table of chemical elements of Mendeleev (who invented the periodic table). Now, compute the sum of products of number of atoms to the atomic weight: Molar mass (Zn (m)) Count i Weight i. 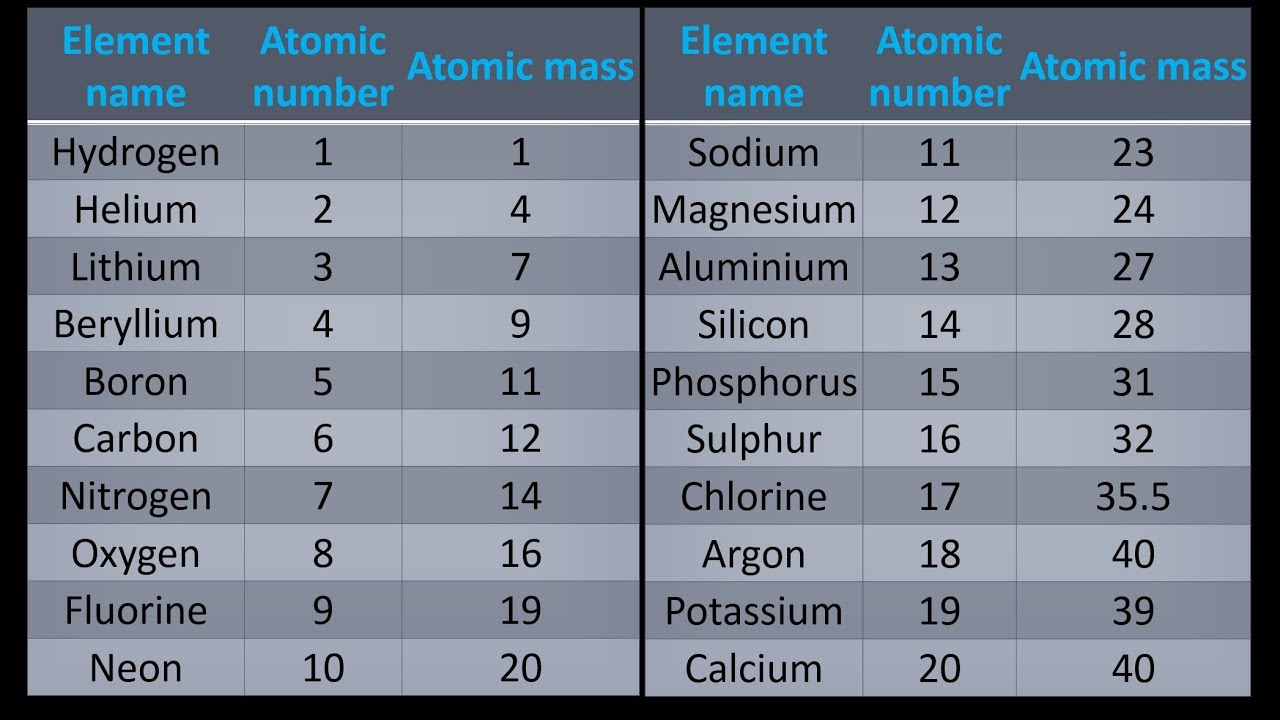
Then, lookup atomic weights for each element in periodic table: Zn: 65.38. Find a periodic table of elements to find the molar mass of your sample. First, compute the number of each atom in Zn (m): Zn: 1. Phone: +31 152 610 900 chemical element contains a link to a page that explains its chemical properties, health effects, environmental effects, application data, an image and also information of the history/inventor of each element. For example, take the example of zinc nitrate, or Zn (NO 3) 2. :max_bytes(150000):strip_icc()/PeriodicTablecolor-58b5d9105f9b586046df7d8a.jpg)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
